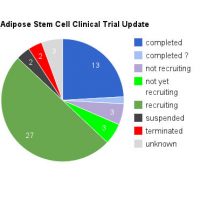
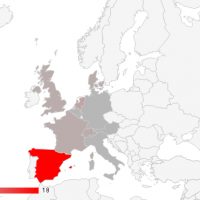
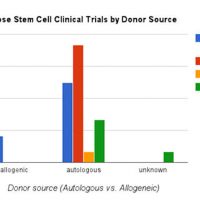
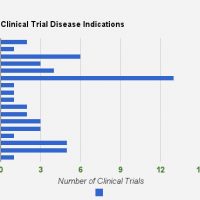
Since I started writing about fat grafts, stem cells, and the regenerative properties and medical potential back in 2009, about 7 years after performing one of my first fat grafting cases, I have become more and more involved and dedicated to the science of fat grafting. As the current President of IFATS (the International Federation for Adipose Therapeutics and Science), I’m excited to be posting a quick note about this year’s conference, which starts tomorrow in San Diego. This year we’re bringing together not only the leading clinical practitioners with whom we are all familiar, but also leading endocrinologists, experts in the field of genomics and big data as applied to medicine, and stem cell researchers from other specialties such as cardiology and orthopedics.