
This year marked the 10th anniversary of IFATS, the International Federation for Adipose Therapeutics and Science. It is an organization which includes scientists and medical doctors from a wide range of specialties. The conference was held in Quebec, Canada and included over 100 abstracts, most of which focused on on adipose stem cells.
Adipose stem cells (ASCs) are a type of adult stem cell which can be easily extracted from your own fat tissue. They have huge application in the treatment of various diseases and injuries. Unlike bone marrow stem cells which have to be culture expanded for several days, ASCs can be isolated and reinjected into the same patient in a few hours time. Due to their ease of extraction and abundant quantities, ASCs are currently being studied in clinical trials and pilot studies for a wide range of diseases. The meeting in Quebec presented abstracts and keynotes from three categories of research:
- Basic science – What’s being done by researchers in labs to study theoretical mechanisms of adipose stem cells
- Translational science – Taking the Basic science and applying it to animals in a lab, or Pilot Studies
- Clinical research – Applying Basic science and Translational Science to humans in clinical trials/case studies
There were many exciting findings, but here are some highlights:
Adipose stem cells (ACS)provide blood supply
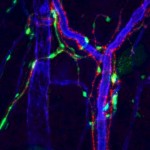
At IFATS, many researchers concluded that adipose stem cells (ASCs) provide vasculature or blood supply to their surrounding environment or tissue. This is important because vascularity promotes tissue healing and skin rejuvenation. Many doctors presenting at the conference reported positive changes in the skin texture and pigmentation of their patients after treatment with ASCs.
The anti-inflammatory properties of adipose stem cells was another topic frequently reported. Researchers concluded that these anti-inflammatory properties are made possible by the cytokines secreted from these stem cells. Cytokines are molecular signals used by cells to communicate or influence cellular behavior. In summary, tissue healing comes from the signals that the cytokines send, rather than cell differentiation.
Animal studies show adipose stem cells are safe

Many labs and investigators reported no adverse events or tumor formations with the use of Adipose Stem Cells (ASCs). The overwhelming consensus is that delivery of ASCs are safe.
In one study, researchers from Indiana University School of Medicine studied the use of ASCs with a mouse model for peripheral arterial disease (PAD). They presented data to show that after administration of freshly isolated stem cells with the tissue genesis cell isolation system, no macroscopic or microscopic forms of tumor formed in mice during a 90 day period.
In another pre-clinical study, University of Indiana Medical School researchers studied Diabetic Retinopathy (DR) in mice. DR is the leading cause of blindness in working age adults. The study successfully demonstrated the safe use of ASC in the treatment of Diabetic Retinopathy. A single intravitreal injection of ASCs limited the features of the Diabetic Retinopathy disease. More study is underway.
Dr. Ra of RNL Bio in Korea concluded that mice can tolerate very high dosages of adipose stem cells. In his study, mice were injected via IV for 13 consecutive weeks with 50 Million cells/kg with no adverse events. It was only in extremely high dosages, 100 Million cells/kg, representing a 10 fold normal dosage, that mice died after the 8th consecutive injection. His lab also compared level of tumor markers from 93 patients who had each received cumulative dosages of 1 Billion cells intravenously (IV) over a period of 18 months. In this group, there was no increase in any tumor markers, aka no cancer. PET scans were also obtained from these patients to show no evidence of cancer or tumor. In a testament to the safety of ASCs, Dr. Ra himself stated that over a 3 year period, he personally had received a total of 8 Billion ASCs.
Clinical trials show safety & efficacy

Currently, there are many Clinical Trials underway with humans to test safety and efficacy of ASCs. To learn more about adipose clinical trials, please see our previous posts.
At IFATS, data was presented from clinical investigators who are studying the use of ASCs in the treatment of Heart Disease, Pulmonary Disease (COPD), Chronic Limb Ischemia (CLI), and Osteoarthritis. No adverse events were reported and there is continued evidence of safety and efficacy. Early results are very exciting!
Ageless Regenerative Institute reported on their clinical trials of both Congestive Heart Failure and Chronic Obstructive Pulmonary Disease (COPD). They collaborated with Bioheart in the heart trial and The Regenerative Medicine Institute of Tijuana, Mexico for both trials.
In the Congestive Heart Failure study, freshly isolated ASC cells from autologous Stromal Vascular Fraction (SVF) were delivered directly into the heart muscle of 15 patients using a catheter. The most recent data showed a average 11% improvement in the EF (ejection fraction) and were able to walk 128 more meters in the ‘6 Minute Walk Test’. To put this in perspective, a football field is approximately 91 meters. Therefore, these patients were walking more than an extra football field length after the treatment. Ejection Fraction (EF) is a measurement of the volume fraction of blood which pumps out of the heart with each beat. An 11% improvement is a big deal.
In the Pulmonary (COPD) study, a total of 34 patients participated. Patients received freshly isolated ASCs from 60 cc of their own fat by IV infusion. This equals a dosage of greater than 30 Million ASCs. At the end of the trial, 83% of the patients demonstrated a statistically significant improvement in quality of life. In addition, at the 3 month mark, these patients were able to walk an additional 7 or more meters in the ‘6 Minute Walk Test’.
Dr. Carlos Garcia presented a case report series for treatment of Lumbar Degenerative Disc Disease (LDDD) with freshly isolated autologous ASCs from SVF. He used 50 cc of adipose tissue. After 6 months, MRI showed increased disc height (recovery) and the patients reported a 90% reduction in pain.
A Korean group, SMGSNU Boramae Medical Center, presented findings from their Phase I and II clinical trial to treat Osteoarthritis in the knee with autologous culture expanded ASCs. Up to 100 Million cells were injected into the knee. Range of motion for all patients improved and MRI’s demonstrated that cartilage was regenerated.
A Phase I Critical Limb Ischemia (CLI) Phase I trials was presented by Dr. Louis Casteilla of StromaLab. Seven patients were injected with autologous culture expanded ASCs. The trial established the endpoint of safety and these patients experienced a significant benefit in wound healing.
Stem cell homing – Cells migrate to the injury
Several investigators presented data to prove the effect of stem cell homing. Homing is the term used when stem cells ‘home’, or find the path to injured organs or damaged tissue. This is most evident when stem cells are administered via IV form. Researchers from Case Western Reserve University induced a pelvic area injury in mice and then injected adipose cells via the tail of the mice. This would be similar to an IV infusion in a human.
In the experiment, the adipose stem cells (ASCs) were tagged, injected into the mice, and then tracked with an MRI to show what part of the body they traveled to. A control group of mice (with no injury) was used to show where ASCs migrate to without any injury. In the injured mice, the ASCs homed to the pelvic injury within 24 hours. These findings support the theory that once injected, stem cells are signaled to ‘home’ to the site of injury to do their magical repair. Also of note is that in the control group of mice with no injury, the ASCs immediately migrated to the lungs.
Method of delivery of ASCs
Researchers from Macquarie Stem Cells in Australia reported on their Clinical Series for Osteoarthritis with 29 patients using autologous ASCs.
Patients were divided into three groups:
- Direct infusion of ASCs into joints
- IV infusion of ASCs (stem cells home)
- IV infusion + direct infusion of ASCs into joints (both combined)
All groups showed improvement. The group with the most amount of improvement was the group treated with both IV infusion and direct infusion into the joints! What was even more amazing was that all patients showed continued improvement in their condition, even after the 6 month period.
ASC cell processing methods & standards

Adipose stem cells are typically isolated from adipose tissue using enzymatic digestion. The entire process can be done in a couple of hours. The fat is extracted by liposuction, decanted, centrifuged, and enzymatically digested with collagenase. The resulting pellet of fat is referred to as the Stromal Vascular Fraction, or SVF. This is the golden nugget of adipose stem cells, a combination of mesenchymal cells and endothelial cells.
Dr. Alexandra Conde Green of JHU reported her findings that the use of enzymes with fat yield much higher stem cell populations compared to fat that is only mechanically processed. Enzyme digestion of adipose tissue yields 10 fold the amount of SVF compared to fat that is only centrifuged (no enzymes). This confirms the importance of enzymes for the highest yield of SVF, but also points to the fact that standard fat grafting (centrifugation only) does contain a small amount of stem cells. Our surgery center collaborated on this paper and Dr. Conde-Greene has written about her stem cell research in our blog.
Mary Pat Moyer, Ph.D. of private company INCELL, presented her findings regarding various methods used to count the number of stem cells in a cc of adipose tissue. During the past few years some commercial companies like Intellicell (SVFC.pk) have been reporting crazy high values for ASC counts per cc of tissue. She concluded that some cell counting meters were greatly exaggerated. As if Intellicell were not in enough trouble already with the FDA, the data presented would push their marketing claims even further under the bus.
Cytori (CYTX), a manufacturer of fat grafting equipment and a point of care ASC processing device, presented values for the average composition of SVF, and estimated the maximum theoretical number of adipose stem cells equal to 4 Million cells/cc. In a jab to exaggerated stem cell count claims by competitors, “anybody reporting cell counts higher than that number is ‘creating life'” . In the Q and A, Dr. Ramon Llull, of GID group, congratulated Cytori in defining this metric. Cytori also presented a methodology to characterize biomarkers from stem cells populations. It is based on microRNA and could help with creating standards and characterization of therapeutic efficacy. This would have direct application for identifying the ‘mechanism of action’ and ‘safety’ in cell populations used in the various clinical trials. Certainly, this is right up the FDA alley.
Dr. Sammy Sliwin, a Canadian surgeon, reported that ASCs separated from lipoaspiratie harvested by water-jet liposuction technique lost properties of stem-ness and therefore might (negatively) affect outcomes of fat grafting procedures.
In a study that compared stem cell counts from fat extracted from different parts of the body, a team from Cooper Medical School concluded that stem cell counts from fat extracted from the flanks, abdomen, and waist were basically equal. However, there was a surprising finding that the ‘arm’ area contained much higher cell counts–a four fold difference!
Dose response curve
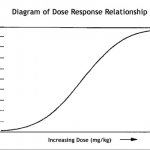
The general finding was that dosage of ASCs matter. At this point in time, the range of IV dosages reported (in humans) was anywhere from .5 Million ASCs to 10 Million ASCs per kg of body weight. Direct injection of 100 Million cells (total) into an injured areas was tolerated well. Many investigators reported highest efficacy with their highest dosage. Likewise, we are in the early stages of plotting a dose response curve for treatments with adipose stem cells.
The dose response curve ultimately defines the number of adipose stem cells required for the treatment of each particular disease or injury.
Who would have ever thought that treating or curing a disease would come down to counting the stem cells from your own unwanted, unloved tissue, known as fat!
Please read our entire Adipose stem cell clinical trial collection
- Part 1: Adipose stem cell clinical trial summary June 2012
- Part 2: Adipose stem cell clinical trials by donor source, Autologous vs. Allogeneic – What’s up with that?
- Part 3: Adipose stem cell clinical trials by country – Spain dominates, Spain rocks at soccer and adipose stem cells
- Part 4: Adipose stem cell clinical trial update – August 2012, 8 trials added




EMILIO MENES says:
Dr. Ricardo L Rodriguez says:
Marilyn Watt says:
Dr. Ricardo L Rodriguez says: