
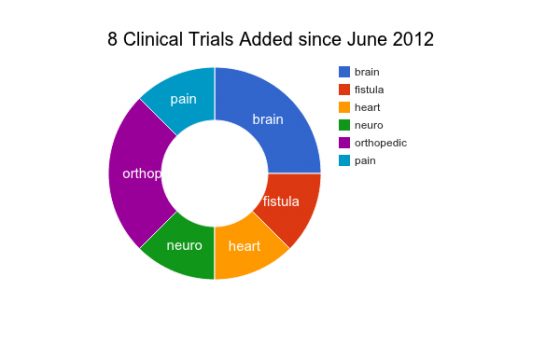
It’s been over two months since I updated my database of Adipose stem cell clinical trials. I am tracking registered clinical trials in the ClinicalTrials.gov database. A lot has happened in only two months! Since June 1, eight new Clinical Trials have been registered or updated to disclose starting dates in the ClinicalTrials.gov database . This brings the all time total of adipose stem cell clinical trials to 62.
Adipose Stem Cells (ASCs) are a type of Adult stem cell and are derived from fat tissue. The common method of extraction to obtain Adipose stem cells involves Liposuction and then isolation of the stem cells by processing the fat with enzymes. The freshly isolated stem cells can be obtained in the same operative session and are referred to as Stromal Vascular Fraction (SVF). Adipose stem cells can also be culture expanded in a GMP lab to yield even higher numbers of cells. However, culture expansion takes days or weeks in a lab.
Conditions under study
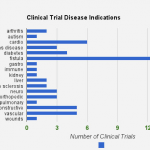
Adipose stem cells are currently being used in clinical trials to treat and cure a wide range of diseases. My original project summary detailed the conditions under study. Conditions include Arthritis, Autism, Crohn’s, Fistula, Kidney, MS, Pulmonary, Diabetes, Cardiac, Neuro, Orthopedic, Reconstructive, and others.
Since June, the conditions with the highest number of new registered clinical trials using adipose stem cells included the brain and orthopedic conditions. Other conditions in the newly registered clinical trial studies include Fistula, Heart, Neuro, and Pain.
All of the new trials are using autologous adipose stem cells, except for the trials studying brain conditions. Clinical trials to study brain conditions are using both autologous and allogeneic Adipose stem cells (ASCs). In Taiwan researchers are studying Severe brain injury with autologous ASCs, but using allogeneic ASCs to treat Cerebellar Ataxia. Autologous stem cells are your own stem cells, and allogeneic stem cells are from another person.
Ways patients receive adipose stem cells
The ways that these adipose stem cells are being delivered to patients include direct injection, intracoronary, and IV infusion. Quite interesting was that both brain trials are using high dosages of adipose stem cells by IV infusion into the bloodstream. The idea is that these stem cells or their cytokines will home to the place of injury–the brain. Many researchers believe that adipose stem cells do cross the blood/brain barrier.
Spain and Korea lead ASC sector
As I outlined in a previous post, Spain is the all time leader of Adipose stem cell clinical trials (at ClinicalTrials.gov) with 18 total Trials. However, during the past two months, Korea is catching up. Of the 8 newly recorded clinical trials, Korea registered a total of 4 trials, with Taiwan, Netherlands, and the USA registering the remaining four trials. Korea now has an all time total of 16 adipose stem cell clinical trials.
If you are just tuning in for the first time, please read the entire Adipose Stem Cell Clinical Trial collection
- Part 1: Adipose stem cell clinical trial update June 2012, Overview with graphs of 54 ASC clinical trials
- Part 2: Adipose stem cell clinical trials by donor source, Autologous vs. Allogeneic – What’s up with that?
- Part 3: Adipose stem cell clinical trials by country – Spain dominates, Spain rocks at soccer and adipose stem cells
- Part 4: You just read it!



